Our work
Central to the understanding of a substance’s toxicity is a consideration of its Absorption, Distribution, Metabolism and Excretion (ADME). How efficiently is a substance absorbed by the human body? How is it broken down? Which tissues does it reach and is it likely to bioaccumulate? How does the body eliminate the substance? All this comes under the general heading of toxicokinetics (TK). Generating new TK data in the laboratory involves the use of animals, is time-consuming and extremely costly (particularly if radiolabels are required). However, bibra specialises in making expert predictions, interpreting ADME assays, and taking into account what is already known about the toxicity of a chemical, its structure, and its physico-chemical properties.
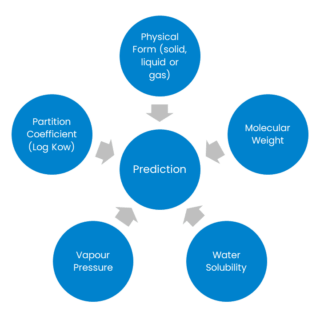
Relevant properties for absorption (and other TK) assessment
In Silico
As well as using physico-chemical properties to predict TK properties, we make use of (Quantitative) Structure-Activity Relationships ((Q)SAR) modelling and software. The US EPA’s EPISuite tool allows physico-chemical properties to be predicted for compounds where experimental data are lacking. There are several “profilers” in the OECD (Q)SAR Toolbox that predict metabolic breakdown products; we have an expert partner who can conduct a similar analysis using the Meteor Nexus software.
TK Assessment under REACH
Conducting new toxicokinetic testing is not a requirement in the EU under the REACH regulation, but above 10 tonnes per year it is necessary to provide an assessment of the existing data. Understanding the toxicokinetic properties of a substance is very useful in determining the most appropriate route of exposure for future testing, likely target organs, and bioaccumulation potential, and in informing on the hazard and risk assessment (e.g. absorption values to be used in DNEL calculations).
Some of our case studies in this area
Determining a Specific Migration Limit (SML) for a data-deficient compound identified in food labels
Case study
The client was informed by a supplier that a chemical compound with a worrying structure was present within a food label ink. Click to find out how we dealt with it.
Pod-based ENDS device PMTA assessments
Case study
As part of a Premarket Tobacco Product Application (PMTA) for a pod-based ENDS device, bibra was asked to provide support preparing non-clinical and human health impact assessments.
ISO 18562 evaluation of a medical device in contact with the breathing gas pathway
Case study
As part of a large programme of work in evaluating the safety of a new endotracheal tube device, bibra was asked to evaluate the health risks posed by exposure to potentially hazardous substances that may be transferred to the patient via the gas stream during device use.
Some industries we work in
REACH
We are a leading supplier of REACH compliance services, assisting chemical manufacturers, importers and downstream users comply with their REACH Regulation responsibilities.

Medical Devices
We are adept at conducting biocompatibility assessments and biological safety evaluations of medical devices in compliance with ISO 10993, FDA and European guidelines, to help bring your device to market.

Pharmaceuticals
Our team is adept at preparing critical toxicological reports - we regularly provide assessments of novel or unexpected impurities, excipients and other intentionally-added substances, and extractables and leachables.


